Applications of 16S rRNA Sequencing
Medical Microbiology
The utilization of 16S rRNA sequencing holds significant value in pathogen identification and disease diagnosis within the realm of clinical microbiology. This method enables the identification of bacteria and archaea in clinical specimens, offering a crucial means to detect pathogens that might be challenging to culture through traditional methods. An illustrative example of its effectiveness can be observed in the work of Chiu and Miller (2019) where the technique was successfully employed to diagnose infections by detecting bacterial DNA in clinical samples that had previously eluded diagnosis using conventional approaches.
In a separate investigation, Yan et al. (2021) conducted a study analyzing stool samples from individuals with Parkinson's disease (PD) and healthy controls through a combined approach of 16S rRNA gene sequencing and gas chromatography-mass spectrometry (GC-MS). Their findings revealed an elevated abundance of genera like Alistipes, Rikenellaceae_RC9_gut_group, Bifidobacterium, and Parabacteroides in PD patients, contrasted with a reduction in Faecalibacterium levels. These outcomes suggest novel avenues for potential therapeutic interventions in PD by means of dietary modifications and regulation of the gut microbiota.
Environmental Microbiology
In environmental studies, 16S rRNA sequencing is crucial for analyzing microbial diversity and ecological functions. It helps monitor the impact of pollution and other environmental changes on microbial communities. For example, Delgado-Baquerizo et al. conducted a global study of soil microbiomes in urban greenspaces to compare their diversity and composition with nearby natural ecosystems, identify consistent microbial residents, evaluate influencing socioeconomic and environmental factors, and assess microbial functional attributes. This work advances our understanding of soil organisms' roles in urban environments and supports sustainable urban greenspace development. Researchers analyzed soil microbial communities using ribosomal DNA analysis to understand their global distribution and response to environmental changes. Findings showed high bacterial diversity and stress-induced variations in community composition. This research informs soil conservation and management strategies amidst climate change and enhances our predictive capabilities for microbial community dynamics in changing environments (Dargiri et al., 2023).
3. Agriculture
In agriculture, 16S rRNA sequencing helps understand the soil microbiome and its impact on crop health and yield. For instance, Fan et al. employed high-throughput sequencing to investigate soil microorganisms for biological control of Fusarium wilt in bananas. Soil microbial diversity analysis in Yunnan's banana-producing areas revealed significant enrichment of Bacillus, negatively correlated with the pathogen Foc TR4. Isolation of Bacillus velezensis strain YN1910 from disease-suppressive soils demonstrated significant control (78.43–81.76%) of banana Fusarium wilt and growth promotion in banana plants in both in vitro and pot experiments. Another study by Bulgarelli et al. (2012) explored the root microbiome of Arabidopsis thaliana, offering insights into plant-microbe interactions and their influence on plant health.
4. Industrial Applications
16S rRNA sequencing is a valuable tool in industrial microbiology for optimizing microbial processes utilized in manufacturing. Cao et al. (2016) employed Illumina high-throughput sequencing to investigate microbial communities within an ANAMMOX-UASB reactor treating high-strength wastewater. Remarkably, the reactor reached a stable operational state within a mere 26 days, showcasing a notable nitrogen removal efficiency. The study observed a gradual increase in the abundance of ANAMMOX bacteria over time, with Candidatus Brocadia emerging as the predominant species. The community composition was primarily constituted by Chloroflexi, Bacteroidetes, and Proteobacteria, with the presence of Denitratisoma from the Proteobacteria phylum noted as well. Furthermore, scanning electron microscopy (SEM) images confirmed the presence of a diverse array of microorganisms within the reactor.
In another industrial context within the food industry, researchers conducted an investigation analyzing fermented milk products (FMPs) originating from multiple regions of Russia. Employing 16S rRNA gene sequencing, the study focused on characterizing the bacterial communities present in these products. The results unveiled that Firmicutes, encompassing genera such as Lactococcus, Lactobacillus, Streptococcus, Lentilactobacillus, and Leuconostoc, were the prevailing taxa, followed by Proteobacteria. Notably, the abundance of Lactobacillus and Lactococcus was particularly prominent in FMPs crafted using raw milk and subject to fermentation at room temperature. Conversely, products produced from pasteurized milk and fermented at higher temperatures were predominantly enriched with Streptococcus. The comparative analysis across different FMPs illuminated both shared microbial profiles and distinctive regional specificities, hinting at promising prospects for innovative product development and enhanced characteristics (Kochetkova et al., 2022).
5. Animal Health
In veterinary science, 16S rRNA sequencing investigates the microbiomes of different animal species, shedding light on their roles in nutrition, immune function, and disease resistance. For example, Kormas et al. (2014) compared gut prokaryotic communities in wild, organically-, and conventionally reared sea bream using 16S rRNA gene sequencing. The number of bacterial OTUs decreased from wild to conventionally reared fish. This study highlights diet-induced changes in sea bream gut microbiota. Another study by Suchodolski (2011) focused on the gut microbiota of dogs and cats, providing valuable information for developing probiotics and other microbiome-targeted therapies.
6. Forensic Science
Forensic applications of 16S rRNA sequencing include microbial forensics, where microbial signatures at crime scenes can be used to link suspects to locations or objects. Emmons et al. (2020) studied microbial colonization of human bone and tooth samples to understand postmortem skeletal degradation. They identified dominant bacterial and eukaryotic colonizers, including Proteobacteria and Ascomycota. However, bacterial loading did not significantly correlate with human DNA concentration, indicating complex interactions between microbial communities and DNA preservation. Similarly, Lax et al. (2015) utilized 16S rRNA sequencing to explore the microbial "fingerprints" left on surfaces, aiding forensic identification.


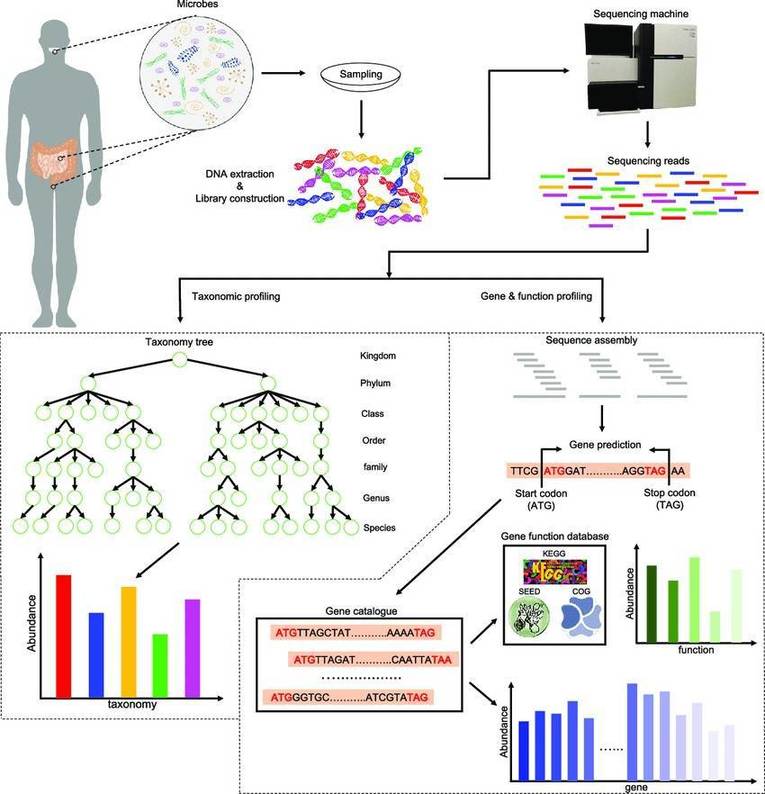
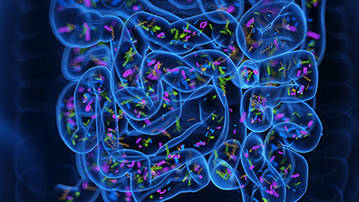
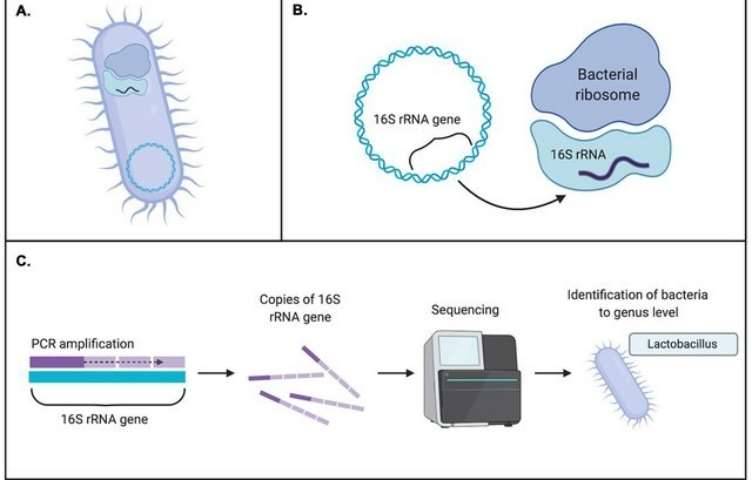 Figure 1. 16S rRNA sequencing is the most common method used to analyze the microbiome. (Patel et al., 2022)
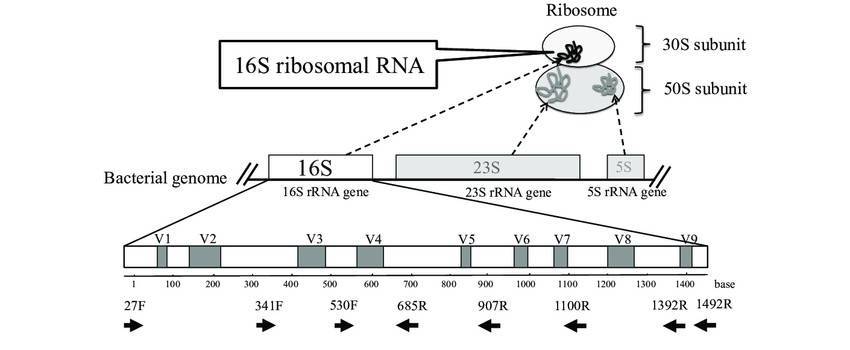
Figure 1. 16S rRNA sequencing is the most common method used to analyze the microbiome. (Patel et al., 2022)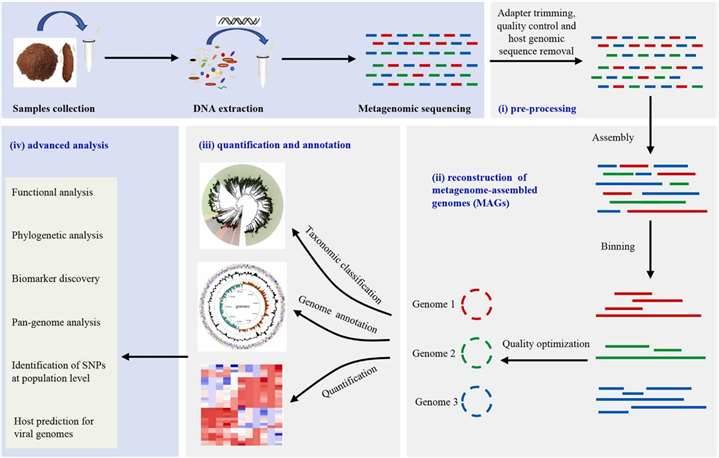 Figure 2. Typical workflow for reconstructing and analyzing metagenome-assembled genomes (MAGs) from metagenomic sequencing data. (Zhou et al., 2022)
Figure 2. Typical workflow for reconstructing and analyzing metagenome-assembled genomes (MAGs) from metagenomic sequencing data. (Zhou et al., 2022)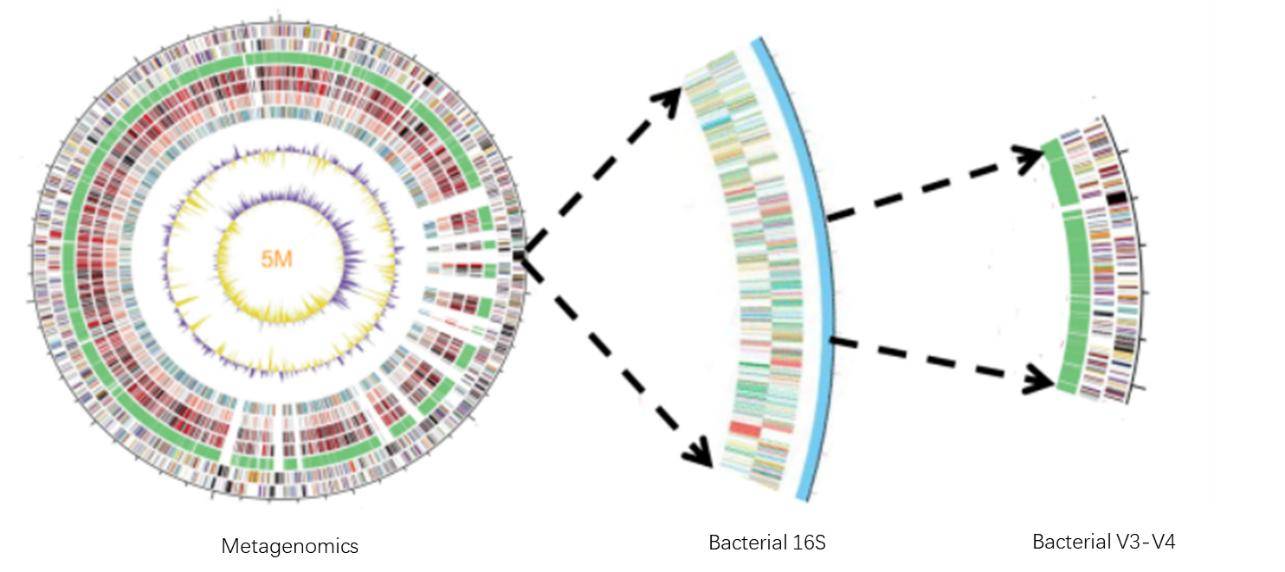 Figure 3. Comparison of Target Regions in Metagenomics and 16S Sequencing.
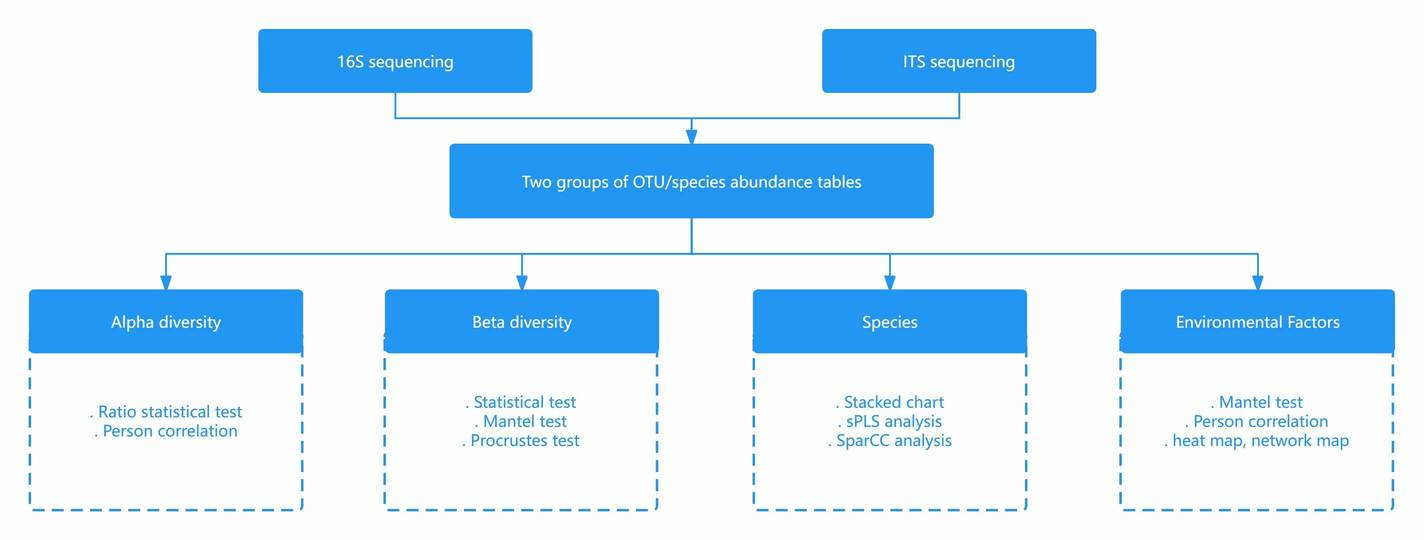
Figure 3. Comparison of Target Regions in Metagenomics and 16S Sequencing.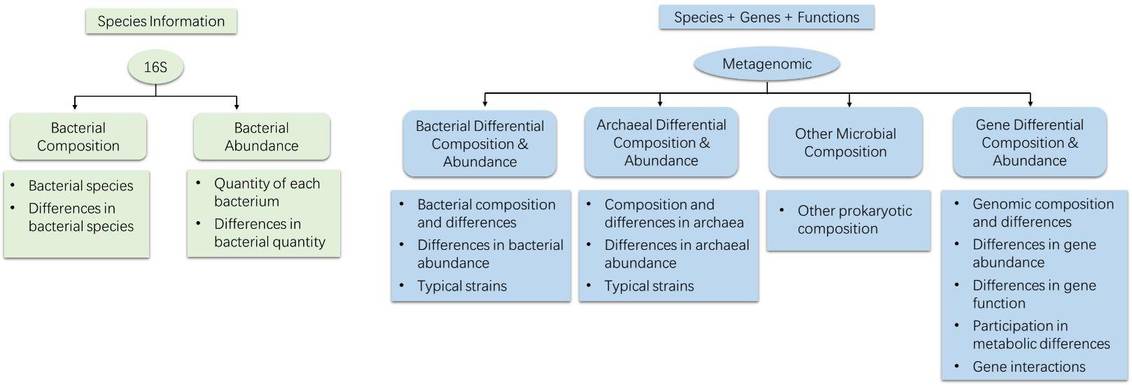 Figure 4. Comparison of Analytical Content in Metagenomics and 16S Sequencing.
Figure 4. Comparison of Analytical Content in Metagenomics and 16S Sequencing.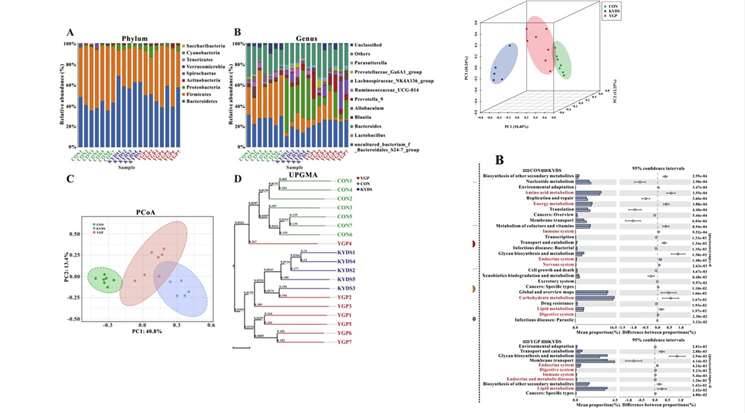 Figure 5. Case Studies of Analytical Results from 16S Sequencing. (Chen et al., 2019)
Figure 5. Case Studies of Analytical Results from 16S Sequencing. (Chen et al., 2019)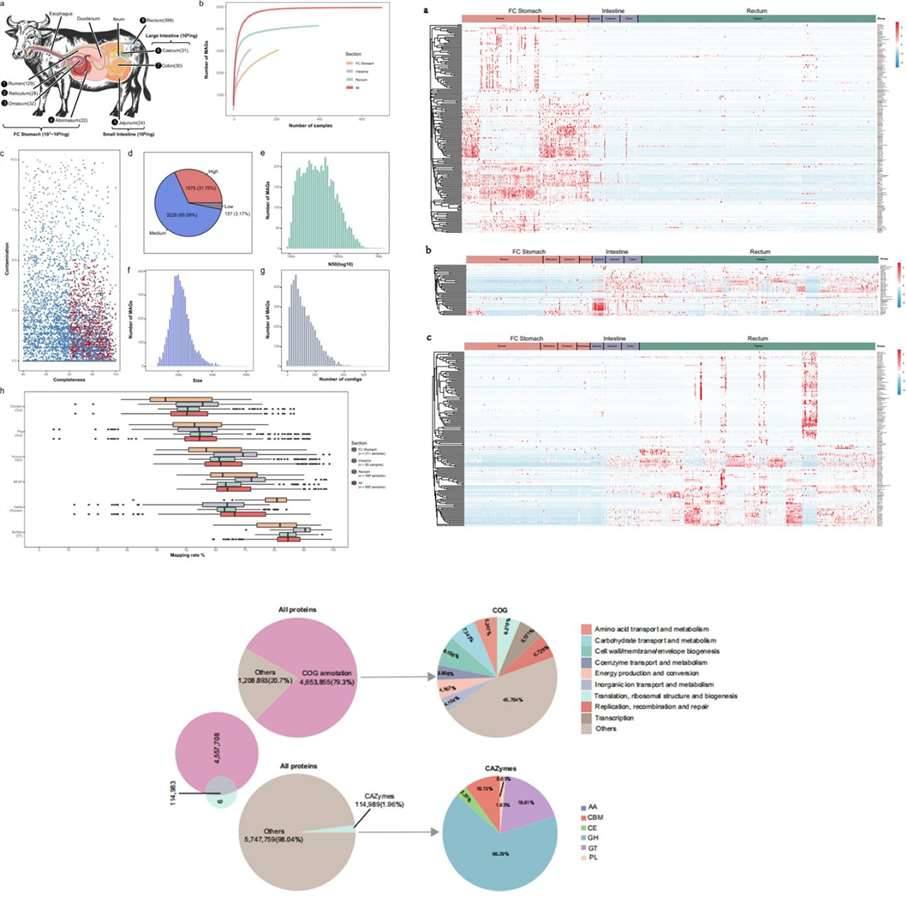 Figure 6. Case Studies of Analytical Results from Metagenomic Sequencing. (Tong et al., 2022)
Figure 6. Case Studies of Analytical Results from Metagenomic Sequencing. (Tong et al., 2022)