The Plant Total RNA Kit is specifically designed for total RNA isolation from plant samples in a fast, simple, and cheap manner. The RNase-free Filtration Column CS in the kit is used to homogenize and filter viscous plants or fungal lysates. Proteins and genomic DNA contamination from plane tissues are removed using a unique silica-membrane technology, a special buffer solution, and a unique DNase I. Total RNA that has been purified can be used directly in molecular biology experiments such as RT-PCR, Real-time RT-PCR, Northern Blot, and RNA library construction. If the sample contains a lot of secondary metabolisms, buffer HL can be used to increase purification efficiency. Fresh or frozen plant species and tissues in amounts of 50-100mg are acceptable sample types.
Storage:
DnaseⅠ, Buffer RDD and RNase-Free ddH2O (tubular) should be stored at 2-8℃. Buffer RL/β-mercaptoethanol mix can be stored at 4°C for 1 month. Other reagents can be stored at room temperature (15-25℃).
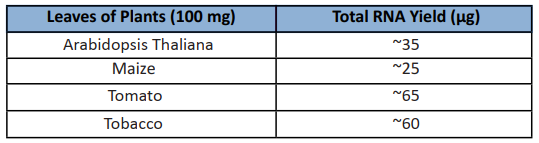
Components:
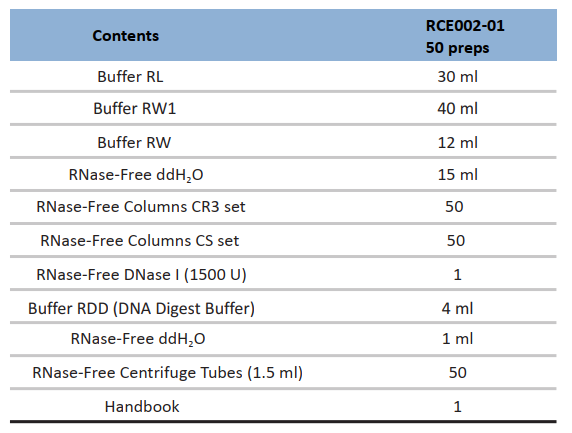
Yield:
Specifications:
| Sample amount | 50-100mg |
| Features | Optimized buffers for plant tissue samples enables a fast and convenient workflow for total RNA isolation within just 40min. Unique filtration column CS eliminates other contaminations. No phenol-chloroform extraction enables the workflow more safety. |
| Application | The purified total RNA can be used directly for molecular biological experiments such as RT-PCR, Real-time RT-PCR, Northern Blot, and RNA library construction, etc. |
| Species Category | plants |
| Sample type | fresh or frozen plant species and tissues |
- Place the 50-100 mg tissue in liquid nitrogen immediately, and grind thoroughly with a mortar and pestle. Add 450 μl Buffer RL (Ensure that β-ME has been added to Buffer RL before use) to the maximum of 100 mg tissue powder. Vortex vigorously.
- Transfer the lysate to RNase-Free Filter Column CS placed in a 2 ml Collection Tube, and centrifuge for 2-5 min at 12,000 rpm (~13,400 × g). Carefully transfer the supernatant to a new centrifuge tube (not supplied) without disturbing the cell-debris pellet in the Collection Tube. Use only this supernatant in subsequent steps.
- Add 0.5 volume of ethanol (96-100%) to the cleared lysate, and mix immediately by pipetting. Transfer the sample, including any precipitate that may have formed, to RNase-Free Spin Column CR3 placed in a 2 ml Collection Tube. Close the lid gently, and centrifuge for 30-60 s at 12,000 rpm (~13,400 × g). Discard the flow-through.
- Add 350 μl Buffer RW1 to the Spin Column CR3. Close the lid gently, and centrifuge for 30-60 s at 12,000 rpm (~13,400 × g). Discard the flow-through.
- Preparation of DNase I working solution: Add 10 μl DNase I stock solution (see Preparation of DNase I stock solution) to 70 μl Buffer RDD. Mix by gently inverting the tube.
- Add the DNase I working solution (80 μl) directly to the center of Spin Column CR3, and place on the bench top (15-25°C) for 15 min.
- Add 350 μl Buffer RW1 to the Spin Column CR3. Close the lid gently, and centrifuge for 30-60 s at 12,000 rpm (~13,400 × g). Discard the flow-through.
- Add 500 μl Buffer RW to the Spin Column CR3 (Ensure that ethanol has been added to Buffer RW before use). Close the lid gently, place in room temperature for 2 min and centrifuge for 30-60 s at 12,000 rpm (~13,400 × g). Discard the flow-through.
- Repeat step 8.
- Centrifuge for 2 min at 12,000 rpm (~13,400 × g) to dry the spin column membrane.
- Place the Spin Column CR3 in a new 1.5 ml Collection Tube (supplied). Add 30-100 μl RNase-Free water directly to the spin column membrane. Close the lid gently, place in room temperature for 2 min and centrifuge for 2 min at 12,000 rpm (~13,400 × g) to elute the RNA.


